Written by Larry G. Arlian, Marjorie, S. Morgan
Translation and adaptation: Efimov S.T.
This translation is dedicated to readers who really wanted to know about this disease.
annotation
Scabies is one of the earliest human diseases, the causes of which have been known from the very beginning. This disease is caused by the itch mite Sarcoptes scabiei , which creates passages and burrows in the epidermis of the skin of humans and many other mammals. Previously, this mite was known under the specific name Acarus scabiei (DeGeer, 1778), but it received its current name since the founding of the genus Sarcoptes (Latreille 1802) in 1802. Research over the past 40 years has greatly expanded our understanding of the biology of this tick species, as expressed in the interaction of the parasite with the host and in the mechanisms that this tick uses to evade the host's defense. This review highlights some of the major advances in our knowledge of biology, the genome, the set of proteins in the parasite's body produced by the tick at a given time ( proteome ), and the immunomodulatory abilities of ticks that provide the basis for disease control. Advances in the development of a blood diagnostic test to detect scabies infection and the emergence of a vaccine to protect susceptible populations from infection, or at least limit transmission of the disease, are also presented.
Origin.
Sarcoptes scabiei, , . , S. scabiei - . , , . , IT . , , https://habr.com/ru/post/537708/.
.
, [ 1 ] [ 2 ] 1900- . , , (1200 . ..) [ 1 ]. [ 2 ], - Acarus scabiei ( Sarcoptes scabiei) , 1687 . , . - , , , .
— , .
Sarcoptes scabiei Acarus Acarus scabiei DeGeer, 1778. S. scabiei. Sarcoptes scabiei Sarcoptoidea Sarcoptidae . S. scabiei Acariformes, Sarcoptiformes, Oribatida, Desmonomata Astigmata ( Dermatophagoides farinae , D. pteronyssinus Euroglyphus maynei ) [ 3 ].
Sarcoptidae (Sarcoptinae, Teinocoptinae Diabolicoptinae), 16 118 , [ 4 , 5 ]. Sarcoptinae Sarcoptes (1 ), Prosarcoptes (3 ), Trixacarus (3 ) Kutzerocoptes (1 ). Sarcoptes, Trixacarus caviae , . Trixacarus caviae - , , Sarcoptes [ 6 ]. Trixacarus caviae , [ 7 ]. , Sarcoptes T. caviae . T. caviae , S. scabiei , T. caviae , S. scabiei, [ 7 ]. – , () () (). sci l1 , d1 T. Caviae, S. scabiei (. 1).
1
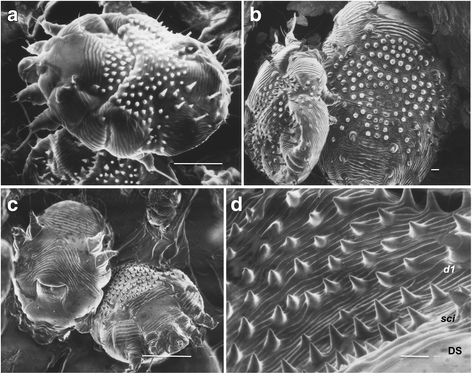
S. scabiei [ 2 , 4 , 5 , 6 ]. , S. scabiei (), (. 1 ). (l) (d) , - . sci, l1 d1 (. 1).
(. 1 , 2 ). III IV - , I II , ( ), (. 2 ). . IV , . (III IV III ) . (. 2). I, II, III IV . I, II III IV.
. 2
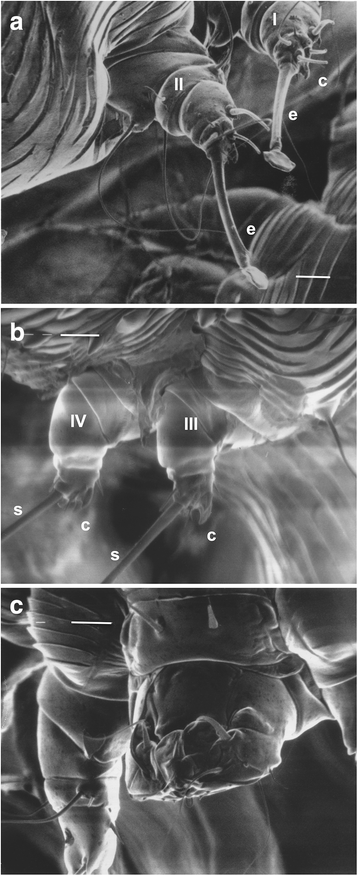
(capitalum) (. 2c ). / , («») (. 3 ). 5,62 ± 1,25 2,8 ± 0,86 [ 8 ]. : 1,49 ± 0,59 0,39 ± 0,16 [ 8 ].
. 3
S. scabiei var. hominis . ( ) , . , ( , , ) , -, . , var. canis var. Hominis .
(-), , . / . 30 . / [ 9]. , , , ( / ).
, , . , ( « »). , , , , , . , , (var. Suis ) - [ 10 ].
S. scabiei , , , . . S. scabiei var. hominis. 12 17 [ 11 ], 17 21 [ 12 ], 7 10 [ 13 ], 9 15 [ 14 ] 15 [ 2]. ]. , 40–50 26–40 [ 2 ].
S. scabiei var. In vivo . canis , 10 13 [ 15 ]. 50–53 . 3–4 , 2–3 . 10–15 var. suis [ 16 ].
S. scabiei . , , in vivo ( , ; in vitro , ). , , , .
Sarcoptes scabiei , , , . , . , , S. Scabiei . , , . Sarcoptes scabiei var. hominis [ 17 ]. , .
, var. canis , , , ( 1 , ) , [ 18 ]. 68% 4,9 (1,93 ) , 100% 4,2 (1,65 ). 20% 11,2 (4,41 ). , . , , , / CO 2 .
, [ 18]. 83% 5,6 (2,2 ). ( 50% ) , , (2,5 = 1 ) . 6,5 (4,41 ) 38% , 5% 32 °C 57% , , , , , . , , , CO2, CO2 .
Mellanby et al. [ 19 ] , S. scabiei var. hominis, 20 ° C 30 ° C, . Sarcoptes scabiei var. hominis, 24 ° C, . 24 ° C.
, (, ) .
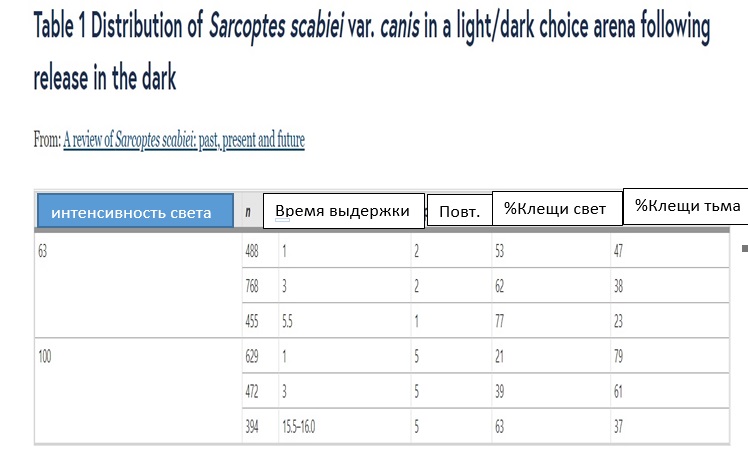
. . Sarcoptes scabiei var. hominis ( ) , [ 19 ]. , S. scabiei var. canis 63 (678 / 2 ) 100 (1076 / 2 ) ( 1). , - , , , .
1 Sarcoptes scabiei var. canis /
. [ 11 ], , 272 4 . - . .
S. scabiei . Arlian et al. [ 20 ] , S. scabiei var. canis 15 ° C (59 ° F) (RH) 75% (. 4 ). 25 ° C (77 ° F) 1-2 RH (. 4).). , . , . - ( ). 20 ° C .
. 4
![The observed time to 100% mortality in the tested female populations of S. scabiei var. canis that have been exposed to certain temperature fluctuations with relative humidity (RH). The number of ticks in each test group ranged from 8 to 26. Data from [20] The observed time to 100% mortality in the tested female populations of S. scabiei var. canis that have been exposed to certain temperature fluctuations with relative humidity (RH). The number of ticks in each test group ranged from 8 to 26. Data from [20]](https://habrastorage.org/getpro/habr/upload_files/c2e/d68/10e/c2ed6810e23de0fb339474c9b2a83936.png)
, S. scabiei var. hominis 19 10 ° C 97%, 8 10 ° C 25% [ 20 ]. Sarcoptes scabiei var. hominis , 12- 4 ° C 10 ° C 95% (21 ° C 45% RH) 4 , [ 20 ].
, , . var. canis -25 ° C 50% 1,5 100% . 1 23% , [ 20 ]. , -15 ° C -17 ° C, .. .
Mellanby et al. [ 19 ] . 10 30 0–90%. 49 ° C (120 ° F) 10 47,5 ° C (117,5 ° F) 30 . - , 84,7, 30,5, 6,8, 1,7 0% 1, 2, 3, 4 5 , , 21,0–25,5 ° C 90%. 24–25 ° C 30% 63,5, 6,8 0% 1, 2 3 , . 61, 52, 35, 30 26% 5, 6, 7, 9 11 90%, 14 . , 4 2% 2 4 30%. , . . 2 0 ° C, 28,6% 8 .
.
c .
1.
Roncalli RA. The history of scabies in veterinary and human medicine from biblical to modern times. Vet Parasitol. 1987;25(2):193–8.
CAS PubMed Article Google Scholar
2.
Friedman R. The story of scabies. New York: Froben Press; 1947.
Google Scholar
3.
Zhang ZQ. Animal biodiversity: an outline of higher-level classification and survey of taxonomic richness. Zootaxa. 2011;3148:237.
Google Scholar
4.
Bochkov AV. A review of mammal-associated Psoroptidia (Acariformes: Astigmata). Acarina. 2010;18:99–260.
Google Scholar
5.
Klompen H. Phylogenetic relationships in the mite family Sarcoptidae (Acari: Astigmata). Misc Publ Univ Michigan Mus Zool. 1992;180:1–155.
Google Scholar
6.
Fain A. Etude de la variabilite de Sarcoptes scabiei avec une revision des Sarcoptidae. Acta Zool Pathol Antverp. 1968;47(1):1–196.
Google Scholar
7.
Kummel BA, Estes SA, Arlian LG. Trixacarus caviae infestation of guinea pigs. J Am Vet Med Assoc. 1980;177(9):903–8.
CAS PubMed Google Scholar
8.
Arlian LG, Runyan RA, Vyszenski-Moher DL. Water balance and nutrient procurement of Sarcoptes scabiei var. canis (Acari: Sarcoptidae). J Med Entomol. 1988;25(1):64–8.
CAS PubMed Article Google Scholar
9.
Arlian LG, Runyan RA, Estes SA. Cross infestivity of Sarcoptes scabiei. J Am Acad Dermatol. 1984;10(6):979–86.
CAS PubMed Article Google Scholar
10.
Mounsey K, Ho MF, Kelly A, Willis C, Pasay C, Kemp DJ, et al. A tractable experimental model for study of human and animal scabies. PLoS Negl Trop Dis. 2010;4(7):e756.
PubMed PubMed Central Article CAS Google Scholar
11.
Mellanby K. The development of symptoms, parasitic infection and immunity in human scabies. Parasitology. 1944;35(4):197.
Article Google Scholar
12.
Heilesen B. Studies on Acarus scabiei and scabies. Rosenkilde & Bagger: Copenhagen; 1946.
Google Scholar
13.
Munro JW. Report of scabies investigation. J R Army Med Corp. 1919;33:1–41.
Google Scholar
14.
Van Neste D, Mrena E, Marchal G. Life cycle of scabies mite (Sarcoptes scabiei¨C11Cvar.¨C12Chominis) studied by scanning electron microscopy (author's transl). Ann Dermatol Venereol. 1981;108(4):355–61.
PubMed Google Scholar
15.
Arlian LG, Vyszenski-Moher DL. Life cycle of¨C13CSarcoptes scabiei¨C14Cvar.¨C15Ccanis. J Parasitol. 1988;74(3):427–30.
CAS PubMed Article Google Scholar
16.
Ljunggren EL. Molecular analysis of¨C16CSarcoptes scabiei. Uppsala: Dept. of Biomedical Sciences and Veterinary Public Health, Swedish University of Agricultural Sciences; 2005.
Google Scholar
17.
Arlian LG, Estes SA, Vyszenski-Moher DL. Prevalence of¨C17CSarcoptes scabiei¨C18Cin the homes and nursing homes of scabietic patients. J Am Acad Dermatol. 1988;19(5,Pt1):806–11.
CAS PubMed Article Google Scholar
18.
Arlian LG, Runyan RA, Sorlie LB, Estes SA. Host-seeking behavior of¨C19CSarcoptes scabiei. J Am Acad Dermatol. 1984;11(4,Pt1):594–8.
CAS PubMed Article Google Scholar
19.
Mellanby K, Johnson CG, Bartley WC, Brown P. Experiments on the survival and behavior of the itch mite¨C20CSarcoptes scabiei¨C21CDeG var.¨C22Chominis. Bull Entomol Res. 1942;33:267–71.
20.
Arlian LG, Runyan RA, Achar S, Estes SA. Survival and infectivity of¨C23CSarcoptes scabiei¨C24Cvar.¨C25Ccanis¨C26Cand var.¨C27Chominis. J Am Acad Dermatol. 1984;11(2,Pt1):210–5.
CAS PubMed Article Google Scholar
21.
Wharton GW, Richards AG. Water vapor exchange kinetics in insects and acarines. Annu Rev Entomol. 1978;23:309–28.
CAS Article Google Scholar
22.
Arlian LG, Veselica MM. Reevaluation of the humidity requirements of the house dust mite¨C28CDermatophagoides farinae¨C29C(Acari: Pyroglyphidae). J Med Entomol. 1981;18(4):351–2.
Article Google Scholar
23.
Arlian LG. Water balance and humidity requirements of house dust mites. Exp Appl Acarol. 1992;16:15–35.
CAS PubMed Article Google Scholar
24.
Van Neste D, Lachapelle JM. Host-parasite relationships in hyperkeratotic (Norwegian) scabies: pathological and immunological findings. Br J Dermatol. 1981;105(6):667–78.
PubMed Article Google Scholar
25.
Van Neste D. Intraepidermal localization of scabies mites overlooked? J Am Acad Dermatol. 1984;10(4):676–7.
PubMed Article Google Scholar
26.
Estes SA, Kummel B, Arlian L. Experimental canine scabies in humans. J Am Acad Dermatol. 1983;9(3):397–401.
CAS PubMed Article Google Scholar
27.
Rapp CM, Morgan MS, Arlian LG. Presence of host immunoglobulin in the gut of¨C30CSarcoptes scabiei¨C31C(Acari: Sarcoptidae). J Med Entomol. 2006;43(3):539–42.
CAS PubMed Article Google Scholar
28.
Morgan MS, Arlian LG. Enzymatic activity in extracts of allergy-causing astigmatid mites. J Med Entomol. 2006;43(6):1200–7.
CAS PubMed Article Google Scholar
29.
Arlian LG, Vyszenski-Moher DL. Response of¨C32CSarcoptes scabiei¨C33Cvar.¨C34Ccanis¨C35C(Acari: Sarcoptidae) to lipids of mammalian skin. J Med Entomol. 1995;32(1):34–41.
CAS PubMed Article Google Scholar
30.
Entrekin DL, Oliver JH Jr. Aggregation of the chicken mite,¨C36CDermanyssus gallinae¨C37C(Acari: Dermanyssidae). J Med Entomol. 1982;19(6):671–8.
CAS PubMed Article Google Scholar
31.
Otieno DA, Hassanali A, Obenchain FA, Sternberg AG, R. Identification of guanine as an assembly pheromone of ticks. Insect Sci Appl. 1985;6(3):667–70.
CAS Google Scholar
32.
Sonenshine DE. Tick pheremones. In: Anonymous biology of ticks. New York: Oxford University Press; 1991. p. 331–69.
Google Scholar
33.
Sonenshine DE, Silverstein RM, West JR. Occurrence of sex attractant pheromone, 2,6-dichlorophenol, in relation to age and feeding in American dog tick,¨C38CDermacentor variabilis¨C39C(Say) (Acari: Ixodidae). J Chem Ecol. 1984;10(1):95–100.
34.
Sonenshine DE. Pheromones and other semiochemicals of the acari. Annu Rev Entomol. 1985;30:1–28.
CAS PubMed Article Google Scholar
35.
Arlian LG, Vyszenski-Moher DL. Responses of¨C40CSarcoptes scabiei¨C41C(Acari: Sarcoptidae) to nitrogenous waste and phenolic compounds. J Med Entomol. 1996;33(2):236–43.
CAS PubMed Article Google Scholar
36.
Arlian LG, Morgan MS, Neal JS. Modulation of cytokine expression in human keratinocytes and fibroblasts by extracts of scabies mites. Am J Trop Med Hyg. 2003;69(6):652–6.
PubMed Google Scholar
37.
Mullins JS, Arlian LG, Morgan MS. Extracts of¨C42CSarcoptes scabiei¨C43Cde Geer downmodulate secretion of IL-8 by skin keratinocytes and fibroblasts and of GM-CSF by fibroblasts in the presence of proinflammatory cytokines. J Med Entomol. 2009;46(4):845–51.
CAS PubMed PubMed Central Article Google Scholar
38.
Hajnicka V, Kocakova P, Slavikova M, Slovak M, Gasperik J, Fuchsberger N, et al. Anti-interleukin-8 activity of tick salivary gland extracts. Parasite Immunol. 2001;23(9):483–9.
CAS PubMed Article Google Scholar
39.
Deruaz M, Frauenschuh A, Alessandri AL, Dias JM, Coelho FM, Russo RC, et al. Ticks produce highly selective chemokine binding proteins with antiinflammatory activity. J Exp Med. 2008;205(9):2019–31.
CAS PubMed PubMed Central Article Google Scholar
40.
Vancova I, Hajnicka V, Slovak M, Kocakova P, Paesen GC, Nuttall PA. Evasin-3-like anti-chemokine activity in salivary gland extracts of ixodid ticks during blood-feeding: a new target for tick control. Parasite Immunol. 2010;32(6):460–3.
CAS PubMed Article Google Scholar
41.
Morgan MS, Arlian LG. Response of human skin equivalents to¨C44CSarcoptes scabiei. J Med Entomol. 2010;47:877–83.
CAS PubMed PubMed Central Article Google Scholar
42.
Morgan MS, Arlian LG, Markey MP.¨C45CSarcoptes scabiei¨C46Cmites modulate gene expression in human skin equivalents. PLoS One. 2013;8(8):e71143.
CAS PubMed PubMed Central Article Google Scholar
43.
Elder BL, Arlian LG, Morgan MS.¨C47CSarcoptes scabiei¨C48C(Acari: Sarcoptidae) mite extract modulates expression of cytokines and adhesion molecules by human dermal microvascular endothelial cells. J Med Entomol. 2006;43(5):910–5.
CAS PubMed PubMed Central Google Scholar
44.
Elder BL, Arlian LG, Morgan MS. Modulation of human dermal microvascular endothelial cells by¨C49CSarcoptes scabiei¨C50Cin combination with proinflammatory cytokines, histamine, and lipid-derived biologic mediators. Cytokine. 2009;47(2):103–11.
CAS PubMed PubMed Central Article Google Scholar
45.
Arlian LG, Morgan MS, Neal JS. Extracts of scabies mites (Sarcoptidae:¨C51CSarcoptes scabiei) modulate cytokine expression by human peripheral blood mononuclear cells and dendritic cells. J Med Entomol. 2004;41(1):69–73.
CAS PubMed Article Google Scholar
46.
Walton SF, Pizzutto S, Slender A, Viberg L, Holt D, Hales BJ, et al. Increased allergic immune response to¨C52CSarcoptes scabiei¨C53Cantigens in crusted¨C54Cversus¨C55Cordinary scabies. Clin Vaccine Immunol. 2010;17(9):1428–38.
CAS PubMed PubMed Central Article Google Scholar
47.
Arlian LG, Morgan MS, Paul CC. Evidence that scabies mites (Acari: Sarcoptidae) influence production of interleukin-10 and the function of T-regulatory cells (Tr1) in humans. J Med Entomol. 2006;43(2):283–7.
CAS PubMed Google Scholar
48.
Holt DC, Fischer K, Allen GE, Wilson D, Wilson P, Slade R, et al. Mechanisms for a novel immune evasion strategy in the scabies mite¨C56CSarcoptes scabiei: a multigene family of inactivated serine proteases. J Invest Dermatol. 2003;121(6):1419–24.
CAS PubMed Article Google Scholar
49.
Bergstrom FC, Reynolds S, Johnstone M, Pike RN, Buckle AM, Kemp DJ, et al. Scabies mite inactivated serine protease paralogs inhibit the human complement system. J Immunol. 2009;182(12):7809–17.
PubMed Article Google Scholar
50.
Fischer K, Langendorf CG, Irving JA, Reynolds S, Willis C, Beckham S, et al. Structural mechanisms of inactivation in scabies mite serine protease paralogues. J Mol Biol. 2009;390(4):635–45.
CAS PubMed Article Google Scholar
51.
Mika A, Reynolds SL, Mohlin FC, Willis C, Swe PM, Pickering DA, et al. Novel scabies mite serpins inhibit the three pathways of the human complement system. PLoS One. 2012;7(7):e40489.
CAS PubMed PubMed Central Article Google Scholar
52.
Mika A, Reynolds SL, Pickering D, McMillan D, Sriprakash KS, Kemp DJ, et al. Complement inhibitors from scabies mites promote streptococcal growth - a novel mechanism in infected epidermis? PLoS Negl Trop Dis. 2012;6(7):e1563.
CAS PubMed PubMed Central Article Google Scholar
53.
Swe PM, Fischer K. A scabies mite serpin interferes with complement-mediated neutrophil functions and promotes staphylococcal growth. PLoS Negl Trop Dis. 2014;8(6):e2928.
PubMed PubMed Central Article Google Scholar
54.
Swe PM, Reynolds SL, Fischer K. Parasitic scabies mites and associated bacteria joining forces against host complement defence. Parasite Immunol. 2014;36((1)1):585–93.
CAS PubMed Article Google Scholar
55.
Arlian LG, Fall N, Morgan MS.¨C57CIn vivo¨C58Cevidence that¨C59CSarcoptes scabiei¨C60C(Acari: Sarcoptidae) is the source of molecules that modulate splenic gene expression. J Med Entomol. 2007;44(6):1054–63.
CAS PubMed Article Google Scholar
56.
Lalli PN, Morgan MS, Arlian LG. Skewed Th1/Th2 immune response to¨C61CSarcoptes scabiei. J Parasitol. 2004;90(4):711–4.
CAS PubMed Article Google Scholar
57.
Rider SD, Morgan MS, Arlian LG. Draft genome of the scabies mite. Parasit Vectors. 2015;8:585.
PubMed PubMed Central Article CAS Google Scholar
58.
Mofiz E, Holt DC, Seemann T, Currie BJ, Fischer K, Papenfuss AT. Genomic resources and draft assemblies of the human and porcine varieties of scabies mites,¨C62CSarcoptes scabiei¨C63Cvar.¨C64Chominis¨C65Cand var.¨C66Csuis. Gigascience. 2016;5(1):23-016-0129-2.
Article CAS Google Scholar
59.
Mofiz E, Seemann T, Bahlo M, Holt D, Currie BJ, Fischer K, et al. Mitochondrial genome sequence of the scabies mite provides insight into the genetic diversity of individual scabies infections. PLoS Negl Trop Dis. 2016;10(2):e0004384.
PubMed PubMed Central Article Google Scholar
60.
Pagel Van Zee J, Geraci NS, Guerrero FD, Wikel SK, Stuart JJ, Nene VM, et al. Tick genomics: the¨C67CIxodes¨C68Cgenome project and beyond. Int J Parasitol. 2007;37((1)2):1297–305.
CAS PubMed Article Google Scholar
61.
Jeyaprakash A, Hoy MA. The nuclear genome of the phytoseiid¨C69CMetaseiulus occidentalis¨C70C(Acari: Phytoseiidae) is among the smallest known in arthropods. Exp Appl Acarol. 2009;47(4):263–73.
CAS PubMed Article Google Scholar
62.
Grbic M, Van Leeuwen T, Clark RM, Rombauts S, Rouze P, Grbic V, et al. The genome of¨C71CTetranychus urticae¨C72Creveals herbivorous pest adaptations. Nature. 2011;479(7374):487–92.
CAS PubMed PubMed Central Article Google Scholar
63.
Chan TF, Ji KM, Yim AK, Liu XY, Zhou JW, Li RQ, et al. The draft genome, transcriptome, and microbiome of¨C73CDermatophagoides farinae¨C74Creveal a broad spectrum of dust mite allergens. J Allergy Clin Immunol. 2015;135(2):539–48.
CAS PubMed Article Google Scholar
64.
Morgan MS, Arlian LG, Rider SD Jr, Grunwald WC Jr, Cool DR. A proteomic analysis of¨C75CSarcoptes scabiei¨C76C(Acari: Sarcoptidae). J Med Entomol. 2016;53(3):553–61.
PubMed PubMed Central Article Google Scholar
65.
Arlian LG, Morgan MS, Rider SD Jr.¨C77CSarcoptes scabiei: genomics to proteomics to biology. Parasit Vectors. 2016;9:380.
PubMed PubMed Central Article Google Scholar
66.
Walton SF, Currie BJ, Kemp DJ. A DNA fingerprinting system for the ectoparasite¨C78CSarcoptes scabiei. Mol Biochem Parasitol. 1997;85(2):187–96.
CAS PubMed Article Google Scholar
67.
Walton SF, Choy JL, Bonson A, Valle A, McBroom J, Taplin D, et al. Genetically distinct dog-derived and human-derived¨C79CSarcoptes scabiei¨C80Cin scabies-endemic communities in northern Australia. Am J Trop Med Hyg. 1999;61(4):542–7.
CAS PubMed Article Google Scholar
68.
Zhao Y, Cao Z, Cheng J, Hu L, Ma J, Yang Y, et al. Population identification of¨C81CSarcoptes hominis¨C82Cand¨C83CSarcoptes canis¨C84Cin China using DNA sequences. Parasitol Res. 2015;114(3):1001–10.
PubMed Article Google Scholar
69.
Zahler M, Essig A, Gothe R, Rinder H. Molecular analyses suggest monospecificity of the genus¨C85CSarcoptes¨C86C(Acari: Sarcoptidae). Int J Parasitol. 1999;29(5):759–66.
CAS PubMed Article Google Scholar
70.
Berrilli F, D'Amelio S, Rossi L. Ribosomal and mitochondrial DNA sequence variation in¨C87CSarcoptes¨C88Cmites from different hosts and geographical regions. Parasitol Res. 2002;88(8):772–7.
CAS PubMed Article Google Scholar
71.
Gu XB, Yang GY. A study on the genetic relationship of mites in the genus¨C89CSarcoptes¨C90C(Acari: Sarcoptidae) in China. Int J Acarol. 2008;34:183–90.
Article Google Scholar
72.
Alasaad S, Soglia D, Spalenza V, Maione S, Soriguer RC, Perez JM, et al. Is ITS-2 rDNA suitable marker for genetic characterization of¨C91CSarcoptes¨C92Cmites from different wild animals in different geographic areas? Vet Parasitol. 2009;159(2):181–5.
CAS PubMed Article Google Scholar
73.
Amer S, El Wahab TA, Metwaly Ael N, Ye J, Roellig D, Feng Y, et al. Preliminary molecular characterizations of¨C93CSarcoptes scabiei¨C94C(Acari: Sarcoptidae) from farm animals in Egypt. PLoS One. 2014;9(4):e94705.
PubMed PubMed Central Article CAS Google Scholar
74.
Walton SF, Dougall A, Pizzutto S, Holt D, Taplin D, Arlian LG, et al. Genetic epidemiology of¨C95CSarcoptes scabiei¨C96C(Acari: Sarcoptidae) in northern Australia. Int J Parasitol. 2004;34(7):839–49.
CAS PubMed Article Google Scholar
75.
Andriantsoanirina V, Ariey F, Izri A, Bernigaud C, Fang F, Guillot J, et al. Wombats acquired scabies from humans and/or dogs from outside Australia. Parasitol Res. 2015;114(6):2079–83.
CAS PubMed Article Google Scholar
76.
Andriantsoanirina V, Ariey F, Izri A, Bernigaud C, Fang F, Charrel R, et al.¨C97CSarcoptes scabiei¨C98Cmites in humans are distributed into three genetically distinct clades. Clin Microbiol Infect. 2015;21(12):1107–14.
CAS PubMed Article Google Scholar
77.
Alasaad S, Soglia D, Sarasa M, Soriguer RC, Perez JM, Granados JE, et al. Skin-scale genetic structure of¨C99CSarcoptes scabiei¨C100Cpopulations from individual hosts: empirical evidence from Iberian ibex-derived mites. Parasitol Res. 2008;104(1):101–5.
CAS PubMed Article Google Scholar
78.
Alasaad S, Oleaga A, Casais R, Rossi L, Min AM, Soriguer RC, et al. Temporal stability in the genetic structure of¨C101CSarcoptes scabiei¨C102Cunder the host-taxon law: empirical evidences from wildlife-derived¨C103CSarcoptes¨C104Cmite in Asturias, Spain. Parasit Vectors. 2011;4:151.
PubMed PubMed Central Article Google Scholar
79.
Alasaad S, Fickel J, Rossi L, Sarasa M, Bena-Tez-Camacho B, Granados JE, et al. Applicability of major histocompatibility complex DRB1 alleles as markers to detect vertebrate hybridization: a case study from Iberian ibex x domestic goat in southern Spain. Acta Vet Scand. 2012;54(1):56.
PubMed PubMed Central Article Google Scholar
80.
Rasero R, Rossi L, Soglia D, Maione S, Sacchi P, Rambozzi L, et al. Host taxon-derived¨C105CSarcoptes¨C106Cmite in European wild animals revealed by microsatellite markers. Biol Conserv. 2010;143(5):1269–77.
Article Google Scholar
81.
Alasaad S, Rossi L, Heukelbach J, Perez JM, Hamarsheh O, Otiende M, et al. The neglected navigating web of the incomprehensibly emerging and re-emerging¨C107CSarcoptes¨C108Cmite. Infect Genet Evol. 2013;17:253–9.
PubMed Article Google Scholar
82.
Renteria-Solis Z, Min AM, Alasaad S, Muller K, Michler FU, Schmaschke R, et al. Genetic epidemiology and pathology of raccoon-derived¨C109CSarcoptes¨C110Cmites from urban areas of Germany. Med Vet Entomol. 2014;28(Suppl 1):98–103.
PubMed Article Google Scholar
83.
Oleaga A, Alasaad S, Rossi L, Casais R, Vicente J, Maione S, et al. Genetic epidemiology of¨C111CSarcoptes scabiei¨C112Cin the Iberian wolf in Asturias, Spain. Vet Parasitol. 2013;196(3–4):453–9.
PubMed Article Google Scholar
84.
Gakuya F, Rossi L, Ombui J, Maingi N, Muchemi G, Ogara W, et al. The curse of the prey:¨C113CSarcoptes¨C114Cmite molecular analysis reveals potential prey-to-predator parasitic infestation in wild animals from Masai Mara, Kenya. Parasit Vectors. 2011;4:193.
PubMed PubMed Central Article Google Scholar
85.
Gakuya F, Ombui J, Maingi N, Muchemi G, Ogara W, Soriguer RC, et al. Sarcoptic mange and cheetah conservation in Masai Mara (Kenya): epidemiological study in a wildlife/livestock system. Parasitology. 2012;139((1)2):1587–95.
PubMed Article Google Scholar
86.
Mellanby K. Scabies. 2nd ed. E. W. Classey: Hampton, UK; 1972.
Google Scholar
87.
Arlian LG, Feldmeier H, Morgan MS. The potential for a blood test for scabies. PLoS Negl Trop Dis. 2015;9:e0004188.
PubMed PubMed Central Article Google Scholar
88.
Arlian LG, Vyszenski-Moher DL, Gilmore AM. Cross-antigenicity between¨C115CSarcoptes scabiei¨C116Cand the house dust mite,¨C117CDermatophagoides farinae¨C118C(Acari: Sarcoptidae and Pyroglyphidae). J Med Entomol. 1988;25(4):240–7.
CAS PubMed Article Google Scholar
89.
Arlian LG, Vyszenski-Moher DL, Ahmed SG, Estes SA. Cross-antigenicity between the scabies mite,¨C119CSarcoptes scabiei, and the house dust mite,¨C120CDermatophagoides pteronyssinus. J Invest Dermatol. 1991;96(3):349–54.
CAS PubMed Article Google Scholar
90.
Arlian LG, Rapp CM, Morgan MS. Resistance and immune response in scabies-infested hosts immunized with¨C121CDermatophagoides¨C122Cmites. Am J Trop Med Hyg. 1995;52(6):539–45.
CAS PubMed Article Google Scholar
91.
Falk ES, Dale S, Bolle R, Haneberg B. Antigens common to scabies and house dust mites. Allergy. 1981;36(4):233–8.
CAS PubMed Article Google Scholar
92.
Walton SF, Slender A, Pizutto S, Mounsey KE, Opresecu F, Thomas WR, et al. Analysis of IgE binding patterns to house dust mite allergens in scabies-endemic communities: insights for both diseases. Clin Exp Allergy. 2015;45((1)2):1868–72.
CAS PubMed Article Google Scholar
93.
Arlian LG, Morgan MS, Estes SA, Walton SF, Kemp DJ, Currie BJ. Circulating IgE in patients with ordinary and crusted scabies. J Med Entomol. 2004;41(1):74–7.
PubMed Article Google Scholar
94.
Arlian LG, Morgan MS, Arends JJ. Immunologic cross-reactivity among various strains of¨C123CSarcoptes scabiei. J Parasitol. 1996;82(1):66–72.
CAS PubMed Article Google Scholar
95.
Hollanders W, Vercruysse J, Raes S, Bornstein S. Evaluation of an enzyme-linked immunosorbent assay (ELISA) for the serological diagnosis of sarcoptic mange in swine. Vet Parasitol. 1997;69(1–2):117–23.
CAS PubMed Article Google Scholar
96.
Kessler E, Matthes HF, Schein E, Wendt M. Detection of antibodies in sera of weaned pigs after contact infection with¨C124CSarcoptes scabiei¨C125Cvar.¨C126Csuis¨C127Cand after treatment with an antiparasitic agent by three different indirect ELISAs. Vet Parasitol. 2003;114(1):63–73.
CAS PubMed Article Google Scholar
97.
Lowenstein M, Kahlbacher H, Peschke R. On the substantial variation in serological responses in pigs to¨C128CSarcoptes scabiei¨C129Cvar.¨C130Csuis¨C131Cusing different commercially available indirect enzyme-linked immunosorbent assays. Parasitol Res. 2004;94(1):24–30.
CAS PubMed Article Google Scholar
98.
van der Heijden HM, Rambags PG, Elbers AR, van Maanen C, Hunneman WA. Validation of ELISAs for the detection of antibodies to¨C132CSarcoptes scabiei¨C133Cin pigs. Vet Parasitol. 2000;89(1–2):95–107.
PubMed Article Google Scholar
99.
Rambozzi L, Menzano A, Lavin S, Rossi L. Biotin-avidin amplified ELISA for detection of antibodies to¨C134CSarcoptes scabiei¨C135Cin chamois (Rupicapra¨C136Cspp.). Vet Res. 2004;35(6):701–8.
100.
Leon-Vizcaino L, Ruiz de Ybanez MR, Cubero MJ, Ortiz JM, Espinosa J, Perez L, et al. Sarcoptic mange in Spanish ibex from Spain. J Wildl Dis. 1999;35(4):647–59.
CAS PubMed Article Google Scholar
101.
Fernandez-Moran J, Gomez S, Ballesteros F, Quiros P, Benito JL, Feliu C, et al. Epizootiology of sarcoptic mange in a population of Cantabrian chamois (Rupicapra pyrenaica parva) in northwestern Spain. Vet Parasitol. 1997;73(1–2):163–71.
CAS PubMed Article Google Scholar
102.
Rodriguez-Cadenas F, Carbajal-Gonzalez MT, Fregeneda-Grandes JM, Aller-Gancedo JM, Huntley JF, Rojo-Vazquez FA. Development and evaluation of an antibody ELISA for sarcoptic mange in sheep and a comparison with the skin-scraping method. Prev Vet Med. 2010;96(1–2):82–92.
CAS PubMed Article Google Scholar
103.
Bornstein S, Frossling J, Naslund K, Zakrisson G, Morner T. Evaluation of a serological test (indirect ELISA) for the diagnosis of sarcoptic mange in red foxes (Vulpes vulpes). Vet Dermatol. 2006;17(6):411–6.
PubMed Article Google Scholar
104.
Curtis CF. Evaluation of a commercially available enzyme-linked immunosorbent assay for the diagnosis of canine sarcoptic mange. Vet Rec. 2001;148(8):238–9.
CAS PubMed Article Google Scholar
105.
Haas N, Wagemann B, Hermes B, Henz BM, Heile C, Schein E. Crossreacting IgG antibodies against fox mite antigens in human scabies. Arch Dermatol Res. 2005;296(7):327–31.
CAS PubMed Article Google Scholar
106.
Oleaga A, Casais R, Gonzalez-Quiros P, Prieto M, Gortazar C. Sarcoptic mange in red deer from Spain: improved surveillance or disease emergence? Vet Parasitol. 2008;154(1–2):103–13.
CAS PubMed Article Google Scholar
107.
Casais R, Prieto M, Balseiro A, Solano P, Parra F, Martin Alonso JM. Identification and heterologous expression of a¨C137CSarcoptes scabiei¨C138CcDNA encoding a structural antigen with immunodiagnostic potential. Vet Res. 2007;38(3):435–50.
CAS PubMed Article Google Scholar
108.
Millan J, Casais R, Delibes-Mateos M, Calvete C, Rouco C, Castro F, et al. Widespread exposure to¨C139CSarcoptes scabiei¨C140Cin wild European rabbits (Oryctolagus cuniculus) in Spain. Vet Parasitol. 2012;183(3–4):323–9.
CAS PubMed Article Google Scholar
109.
He R, Shen N, Lin H, Gu X, Lai W, Peng X, et al. Molecular characterization of calmodulin from¨C141CSarcoptes scabiei. Parasitol Int. 2017;66(2):1–6.
CAS PubMed Article Google Scholar
110.
Mattsson JG, Ljunggren EL, Bergstrom K. Paramyosin from the parasitic mite¨C142CSarcoptes scabiei: cDNA cloning and heterologous expression. Parasitology. 2001;122(Pt 5):555–62.
CAS PubMed Google Scholar
111.
Kuhn C, Lucius R, Matthes HF, Meusel G, Reich B, Kalinna BH. Characterisation of recombinant immunoreactive antigens of the scab mite¨C143CSarcoptes scabiei. Vet Parasitol. 2008;153(3–4):329–37.
CAS PubMed Article Google Scholar
112.
Angelone-Alasaad S, Molinar Min A, Pasquetti M, Alagaili AN, D'Amelio S, Berrilli F, et al. Universal conventional and real-time PCR diagnosis tools for¨C144CSarcoptes scabiei. Parasit Vectors. 2015;8:587.
PubMed PubMed Central Article CAS Google Scholar
113.
Mounsey KE, McCarthy JS. Treatment and control of scabies. Curr Opin Infect Dis. 2013;26(2):133–9.
PubMed Article Google Scholar
114.
Liu X, Walton S, Mounsey K. Vaccine against scabies: necessity and possibility. Parasitology. 2014;141(6):725–32.
PubMed Article Google Scholar
115.
Thomas J, Peterson GM, Walton SF, Carson CF, Naunton M, Baby KE. Scabies: an ancient global disease with a need for new therapies. BMC Infect Dis. 2015;15:250.
PubMed PubMed Central Article Google Scholar
116.
Arlian LG, Morgan MS, Vyszenski-Moher DL, Stemmer BL.¨C145CSarcoptes scabiei: the circulating antibody response and induced immunity to scabies. Exp Parasitol. 1994;78(1):37–50.
CAS PubMed Article Google Scholar
117.
Arlian LG, Morgan MS, Rapp CM, Vyszenski-Moher DL. The development of protective immunity in canine scabies. Vet Parasitol. 1996;62(1–2):133–42.
CAS PubMed Article Google Scholar
118.
Tarigan S, Huntley JF. Failure to protect goats following vaccination with soluble proteins of¨C146CSarcoptes scabiei: evidence for a role for IgE antibody in protection. Vet Parasitol. 2005;133(1):101–9.
CAS PubMed Article Google Scholar
119.
Arlian LG, Rapp CM, Vyszenski-Moher DL, Morgan MS.¨C147CSarcoptes scabiei: histopathological changes associated with acquisition and expression of host immunity to scabies. Exp Parasitol. 1994;78(1):51–63.
CAS PubMed Article Google Scholar
120.
Morgan MS, Arlian LG. Serum antibody profiles of¨C148CSarcoptes scabiei¨C149Cinfested or immunized rabbits. Folia Parasitol (Praha). 1994;41(3):223–7.
CAS Google Scholar
121.
Harumal P, Morgan M, Walton SF, Holt DC, Rode J, Arlian LG, et al. Identification of a homologue of a house dust mite allergen in a cDNA library from¨C150CSarcoptes scabiei¨C151Cvar.¨C152Chominis¨C153Cand evaluation of its vaccine potential in a rabbit/S. scabiei¨C154CVar.¨C155Ccanis¨C156Cmodel. Am J Trop Med Hyg. 2003;68(1):54–60.
CAS PubMed Google Scholar
122.
Casais R, Granda V, Balseiro A, Del Cerro A, Dalton KP, Gonzalez R, et al. Vaccination of rabbits with immunodominant antigens from¨C157CSarcoptes scabiei¨C158Cinduced high levels of humoral responses and pro-inflammatory cytokines but confers limited protection. Parasit Vectors. 2016;9:435.
PubMed PubMed Central Article Google Scholar
123.
Uhlir J. Immunization of rabbits with antigens from¨C159CPsoroptes cuniculi, the rabbit scab mite. Folia Parasitol (Praha). 1992;39(4):375–82.
CAS Google Scholar
124.
Smith WD, Bates P, Pettit DM, Van Den Broek A, Taylor MA. Attempts to immunize sheep against the scab mite,¨C160CPsoroptes ovis. Parasite Immunol. 2002;24(6):303–10.
125.
Smith WD, Pettit DM. Immunization against sheep scab: preliminary identification of fractions of¨C161CPsoroptes ovis¨C162Cwhich confer protective effects. Parasite Immunol. 2004;26(6–7):307–14.
CAS PubMed Article Google Scholar
126.
Gu X, Xie Y, Wang S, Peng X, Lai S, Yang G. Immune response induced by candidate¨C163CSarcoptes scabiei¨C164Cvar.¨C165Ccuniculi¨C166CDNA vaccine encoding paramyosin in mice. Exp Appl Acarol. 2014;63(3):401–12.
CAS PubMed Article Google Scholar
127.
Normaznah Y, Saniah K, Nazma M, Mak JW, Krishnasamy M, Hakim SL. Seroprevalence of¨C167CSarcoptes scabiei¨C168Cvar.¨C169Ccanis¨C170Cantibodies among aborigines in peninsular Malaysia. Southeast Asian J Trop Med Public Health. 1996;27(1):53–6.
CAS PubMed Google Scholar
128.
Raez-Bravo A, Granados JE, Serrano E, Dellamaria D, Casais R, Rossi L, et al. Evaluation of three enzyme-linked immunosorbent assays for sarcoptic mange diagnosis and assessment in the Iberian ibex,¨C171CCapra pyrenaica. Parasit Vectors. 2016;9:558.
PubMed PubMed Central Article Google Scholar
129.
Rodriguez-Cadenas F, Carbajal-Gonzalez MT, Fregeneda-Grandes JM, Aller-Gancedo JM, Rojo-Vazquez FA. Clinical evaluation and antibody responses in sheep after primary and secondary experimental challenges with the mange mite¨C172CSarcoptes scabiei¨C173Cvar.¨C174Covis. Vet Immunol Immunopathol. 2010;133(2–4):109–16.
CAS PubMed Article Google Scholar
130.
Smets K, Vercruysse J. Evaluation of different methods for the diagnosis of scabies in swine. Vet Parasitol. 2000;90(1–2):137–45.
CAS PubMed Article Google Scholar
131.
Vercruysse J, Geurden T, Peelaers I. Development and Bayesian evaluation of an ELISA to detect specific antibodies to¨C175CSarcoptes scabiei¨C176Cvar.¨C177Csuis¨C178Cin the meat juice of pigs. Vet Rec. 2006;158(15):506–8.
CAS PubMed Article Google Scholar
132.
Rampton M, Walton SF, Holt DC, Pasay C, Kelly A, Currie BJ, et al. Antibody responses to¨C179CSarcoptes scabiei¨C180Capolipoprotein in a porcine model: relevance to immunodiagnosis of recent infection. PLoS One. 2013;8(6):e65354.
CAS PubMed PubMed Central Article Google Scholar
133.
Haas C, Rossi S, Meier R, Ryser-Degiorgis MP. Evaluation of a commercial ELISA for the detection of antibodies to¨C181CSarcoptes scabiei¨C182Cin wild boar (Sus scrofa). J Wildl Dis. 2015;51(3):729–33.
CAS PubMed Article Google Scholar
134.
Davidson RK, Bornstein S, Handeland K. Long-term study of¨C183CSarcoptes scabiei¨C184Cinfection in Norwegian red foxes (Vulpes vulpes) indicating host/parasite adaptation. Vet Parasitol. 2008;156(3–4):277–83.
PubMed Article Google Scholar
135.
Jakubek EB, Mattsson R, Morner T, Mattsson JG, Gavier-Widen D. Potential application of serological tests on fluids from carcasses: detection of antibodies against¨C185CToxoplasma gondii¨C186Cand¨C187CSarcoptes scabiei¨C188Cin red foxes (Vulpes vulpes). Acta Vet Scand. 2012;54(1):13.
PubMed PubMed Central Article Google Scholar
136.
Lower KS, Medleau LM, Hnilica K, Bigler B. Evaluation of an enzyme-linked immunosorbent assay (ELISA) for the serological diagnosis of sarcoptic mange in dogs. Vet Dermatol. 2001;12(6):315–20.
CAS PubMed Article Google Scholar
137.
Bornstein S, Zakrisson G. Humoral antibody response to experimental¨C189CSarcoptes scabiei¨C190Cvar.¨C191Cvulpes¨C192Cinfection in the dog. Vet Derm. 1993;4:107–10.
Article Google Scholar
138.
Fuchs B, Zimmermann B, Wabakken P, Bornstein S, Mansson J, Evans AL, et al. Sarcoptic mange in the Scandinavian wolf¨C193CCanis lupus¨C194Cpopulation. BMC Vet Res. 2016;12:156.
PubMed PubMed Central Article Google Scholar
139.
Sarasa M, Rambozzi L, Rossi L, Meneguz PG, Serrano E, Granados JE, et al.¨C195CSarcoptes scabiei: specific immune response to sarcoptic mange in the Iberian ibex¨C196CCapra pyrenaica¨C197Cdepends on previous exposure and sex. Exp Parasitol. 2010;124(3):265–71.
CAS PubMed Article Google Scholar
140.
Bornstein S, Zakrisson G. Clinical picture and antibody response in pigs infected by¨C198CSarcoptes scabiei¨C199Cvar.¨C200Csuis. Vet Derm. 1993;4:123–31.
Article Google Scholar
141.
Zhang R, Jise Q, Zheng W, Ren Y, Nong X, Wu X, et al. Characterization and evaluation of a¨C201CSarcoptes scabiei¨C202Callergen as a candidate vaccine. Parasit Vectors. 2012;5:176.
PubMed PubMed Central Article CAS Google Scholar
142.
Zhang R, Zheng W, Wu X, Jise Q, Ren Y, Nong X, et al. Characterisation and analysis of thioredoxin peroxidase as a potential antigen for the serodiagnosis of sarcoptic mange in rabbits by dot-ELISA. BMC Infect Dis. 2013;13:336.
CAS PubMed PubMed Central Article Google Scholar
143.
Zheng Y, He R, He M, Gu X, Wang T, Lai W, et al. Characterization of¨C203CSarcoptes scabiei¨C204Ccofilin gene and assessment of recombinant cofilin protein as an antigen in indirect-ELISA for diagnosis. BMC Infect Dis. 2016;16:21.
PubMed PubMed Central Article CAS Google Scholar
144.
Jayaraj R, Hales B, Viberg L, Pizzuto S, Holt D, Rolland JM, et al. A diagnostic test for scabies: IgE specificity for a recombinant allergen of¨C205CSarcoptes scabiei. Diagn Microbiol Infect Dis. 2011;71(4):403–7.
CAS PubMed Article Google Scholar
145.
Casais R, Goyena E, Martinez-Carrasco C, Ruiz de Ybanez R, Alonso de Vega F, Ramis G, et al. Variable performance of a human derived¨C206CSarcoptes scabiei¨C207Crecombinant antigen ELISA in swine mange diagnosis. Vet Parasitol. 2013;197(1–2):397–403.
CAS PubMed Article Google Scholar
146.
Casais R, Millan J, Rosell JM, Dalton KP, Prieto JM. Evaluation of an ELISA using recombinant Ssλ20ΔB3 antigen for the serological diagnosis of¨C208CSarcoptes scabiei¨C209Cinfestation in domestic and wild rabbits. Vet Parasitol. 2015;214(3–4):315–21.
CAS PubMed Article Google Scholar
147.
Oleaga A, Casais R, Balseiro A, Espi A, Llaneza L, Hartasanchez A, et al. New techniques for an old disease: sarcoptic mange in the Iberian wolf. Vet Parasitol. 2011;181(2–4):255–66.
PubMed Article Google Scholar
148.
Dougall A, Holt DC, Fischer K, Currie BJ, Kemp DJ, Walton SF. Identification and characterization of¨C210C Sarcoptes scabiei ¨C211Cand¨C212C Dermatophagoides pteronyssinus ¨C213Cglutathione S-transferases: implication as a potential major allergen in crusted scabies. Am J Trop Med Hyg. 2005; 73 (5): 977–84.